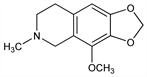
4788. Hydrocotarnine
Nomenclature
CAS number: 550-10-7
5,6,7,8-Tetrahydro-4-methoxy-6-methyl-1,3-dioxolo[4,5-g]isoquinoline; 8-methoxy-5,6-methylenedioxy-2-methyl-1,2,3,4-tetrahydroisoquinoline. C12H15NO3; mol wt 221.25.
C 65.14%, H 6.83%, N 6.33%, O 21.69%.
Description and references
Found in mother liquors from morphine extraction. It is not certain whether it is formed from narcotine during the extraction or whether it exists in the poppy plant. May also be prepd by reduction of cotarnine: Topchiev, J. Appl. Chem. USSR 6, 529 (1933), C.A. 28, 2718 (1934); Schneider, Müller, Ann. 615, 34 (1958); Knabe, Arch. Pharm. 292, 652 (1959). Reduction of hydrocotarnine with sodium in alcohol leads to replacement of the methoxyl group by hydrogen, with formation of hydrohydrastinine. Review and bibliography: Small, Lutz, “Chemistry of the Opium Alkaloids,” Suppl. No. 103, Public Health Reports, Washington (1932).