4787. Hydrocortisone
Nomenclature
Description and references
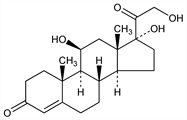
Principal glucocorticoid hormone produced by the adrenal cortex. Biosynthesis stimulated by ACTH, q.v. Circulates in plasma primarily bound to corticosteroid-binding globulin, also known as transcortin, and to albumin. Isoln from adrenal glands: Reichstein, Helv. Chim. Acta 20, 953 (1937); Mason et al., J. Biol. Chem. 124, 459 (1938); from urine: Mason, Sprague, ibid. 175, 451 (1948); from blood: Reich et al., ibid. 187, 411 (1950). Configuration: von Euw, Reichstein, Helv. Chim. Acta 25, 988 (1942); 30, 205 (1947). Synthesis: N. L. Wendler et al., J. Am. Chem. Soc. 72, 5793 (1950). Biosynthesis by isolated adrenal glands: O. Hechter et al., Arch. Biochem. Biophys. 25, 457 (1950); A. Zaffaroni et al., J. Am. Chem. Soc. 73, 1390 (1951). Prepn by microbial transformation: H. C. Murray, D. H. Peterson, US 2602769 (1952 to Upjohn); D. R. Colingsworth et al., J. Biol. Chem. 203, 807 (1953). Total biosynthesis in yeast: F. M. Szczebara et al., Nat. Biotechnol. 21, 143 (2003). Comprehensive description: K. Florey, Anal. Profiles Drug Subs. 12, 277-324 (1983). Review of clinical use in dermatoses: A. M. Kligman, K. H. Kaidbey, Cutis 22, 232-244 (1978). Review of clinical assays in serum and urine: A. Moore et al., Ann. Clin. Biochem. 22, 435-454 (1985). Physiological role in immunity: W. M. Jefferies, Med. Hypotheses 34, 198-208 (1991); in fetal maturation: G. C. Liggins, Reprod. Fertil. Dev. 6, 141-150 (1994).
Properties
Bitter-tasting crystalline, striated blocks from abs ethanol or isopropanol, mp 217-220° with some decompn. [α]D22 +167° (abs ethanol). uv max: 242 nm (E1%1cm 445). Soly (mg/ml) at 25°: water 0.28; ethanol 15.0; methanol 6.2; acetone 9.3; chloroform 1.6; propylene glycol 12.7; ether about 0.35. Sol in concd sulfuric acid with intense green fluorescence.Derivative
21-Acetate.Nomenclature
Properties
Monoclinic, sphenoidal, tabular crystals from dil acetone. Tasteless. d420 1.289; dec 223°. [α]D25 +166° (c = 0.4 in dioxane); [α]D25 +150.7° (c = 0.5 in acetone). uv max (methanol): 242 nm (E1%1cm 390). Somewhat hygroscopic. Soly in water: 1 mg/100 ml; in ethanol: 0.45 g/100 ml; in methanol: 3.9 mg/ml; in acetone: 1.1 mg/g; in ether: 0.15 mg/ml. One gram dissolves in about 200 ml chloroform. Very sol in DMF; sol in dioxane.Derivative
17-Butyrate.Nomenclature
Derivative
21-Phosphate disodium salt.Nomenclature
Properties
White powder. uv max (methanol): 242 nm (A1%1cm 298-341). [α]D25 +120° (H2O). Soly in water (25°): >500 mg/ml. pH of a 1% aq soln: 7.5-8.5.Derivative
21-Sodium succinate.Nomenclature
Properties
Amorphous, hygroscopic, white powder, mp 169.0-171.2°. Soly in water: ≈500 mg/ml. Similarly sol in methanol, ethanol; sparingly sol in chloroform.Derivative
17-Valerate.Nomenclature
Therapeutic Category
Glucocorticoid.
Therapeutic Category (Veterinary)
Glucocorticoid.Keywords
Glucocorticoid