Nomenclature
CAS number: 155-54-4
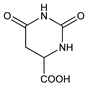
Hexahydro-2,6-dioxo-4-pyrimidinecarboxylic acid; 4,5-dihydroorotic acid.
C
5H
6N
2O
4; mol wt 158.11.
C 37.98%, H 3.82%, N 17.72%, O 40.48%.
Description and references
Prepn from carbethoxyasparagine: Miller et al., J. Am. Chem. Soc. 75, 6086 (1953); US 2773872 (1956 to Merck & Co.).
Derivative
l-Form.
Properties
Crystals, dec 266°. [α]D25.3 +33.23° (c =
1.992 in 1% NaHCO3).Derivative
d-Form.
Properties
Crystals from water, dec 266°. [α]D25.3 31.54° (c = 2.01 in 1% NaHCO3).Derivative
dl-Form.
Properties
Crystals, dec 259°. Forms a water-sol salt.Use
The
l- and
dl-dihydroorotic acids
are precursors to the biological pyrimidines, such as thymine, uracil,
cytosine and hydroxymethylcytosine. When employed in large amounts
d-dihydroorotic acid is an antimetabolite of
l-dihydroorotic
acid in pyrimidine utilization. It has also been found to inhibit
the orotic acid utilization of those organisms which have a requirement
for it in order to grow. In general it may be said to inhibit bacterial
growth.
