4803. Hydromorphone
Nomenclature
CAS number: 466-99-9
(5α)-4,5-Epoxy-3-hydroxy-17-methylmorphinan-6-one; dihydromorphinone. C17H19NO3; mol wt 285.34.
C 71.56%, H 6.71%, N 4.91%, O 16.82%.
Description and references
Semisynthetic opioid analgesic. Prepn from morphine: DE 365683; DE 623821 (1922, 1936 both to Knoll); by oxidation of dihydromorphine: H. Rapoport et al., J. Org. Chem. 15, 1103 (1950). Crystal structure: Steinmetz, Z. Kristallogr. 67, 434 (1928). Pharmacology: N. B. Eddy, J. G. Reid, J. Pharmacol. Exp. Ther. 52, 468 (1934). Toxicity data: M. E. Buchwald, G. S. Eadie, ibid. 71, 197 (1941). GC-MS determn in blood: R. Meatherall, J. Anal. Toxicol. 29, 301 (2005). Review of pharmacology and clinical efficacy: N. Sarhill et al., Support. Care Cancer 9, 84-96 (2001); A. Murray, N. A. Hagen, J. Pain Symptom Manage. 29, S57-S66 (2005).
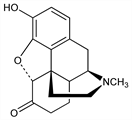
Properties
Crystals from ethanol, mp 266-267°. [α]D25 194° (c = 0.98 in dioxane).Derivative
Hydrochloride.Nomenclature
CAS number: 71-68-1
Dilaudid (Abbott); Opidol (Mundipharma); Palladon (Mundipharma); Palladone (Napp); Sophidone (BMS). C17H19NO3.HCl; mol wt 321.80.
C 63.45%, H 6.26%, N 4.35%, O 14.92%, Cl 11.02%.
Properties
Fine white, or practically white, odorless, crystalline powder, mp 305-315° with decompn (evacuated tube). [α]D25 133.0° (c = 1 in water). Sol in 3 parts water; sparingly sol in alcohol. Practically insol in ether. LD50 in mice (mg/kg): 61-96 i.v. (Buchwald, Eadie). Protect from light.Note
This is a controlled substance (opiate): 21 CFR, 1308.12.Therapeutic Category
Analgesic.
Keywords
Analgesic; Opioids; Phenanthrenes