4818. Hydroxychloroquine
Nomenclature
CAS number: 118-42-3
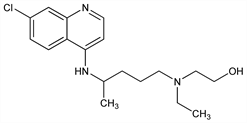
2-[[4-[(7-Chloro-4-quinolinyl)amino]pentyl]ethylamino]ethanol; 7-chloro-4-[4-[ethyl(2-hydroxyethyl)amino]-1-methylbutylamino]quinoline; 7-chloro-4-[4-(N-ethyl-N-β-hydroxyethylamino)-1-methylbutylamino]quinoline; 7-chloro-4-[5-(N-ethyl-N-2-hydroxyethylamino)-2-pentyl]aminoquinoline; oxychloroquine; oxichlorochine. C18H26ClN3O; mol wt 335.87.
C 64.37%, H 7.80%, Cl 10.56%, N 12.51%, O 4.76%.
Description and references
Prepd by reacting a mixture of 4,7-dichloroquinoline, phenol and N′-ethyl-N′-β-hydroxyethyl-1,4-pentadiamine at 125-130°: Surrey, Hammer, J. Am. Chem. Soc. 72, 1814 (1950); Surrey, US 2546658 (1951 to Sterling Drug). Use in combination with cyclophosphamide and azathioprine, q.q.v. in the treatment of rheumatoid arthritis: D. J. McCarty, G. F. Carrera, J. Am. Med. Assoc. 248, 1718 (1982). Reassessment in the treatment of rheumatoid arthritis: Am. J. Med. 75, no. 1A, 1-56 (1983). Series of articles on clinical use: ibid. 85, Suppl. 4A, 1-71 (1988).
Properties
Crystals from ethylene dichloride and Skellysolve B; mp 89-91°.Derivative
Diphosphate.C18H26ClN3O.2H3PO4; mol wt 531.86.
C 40.65%, H 6.06%, Cl 6.67%, N 7.90%, O 27.07%, P 11.65%.
Properties
Recrystallized from ethanol, mp 168-170° (dec).Derivative
Sulfate.Nomenclature
CAS number: 747-36-4
Ercoquin; Plaquenil Sulfate (Winthrop); Quensyl. C18H26ClN3O.H2SO4; mol wt 433.95.
C 49.82%, H 6.50%, Cl 8.17%, N 9.68%, O 18.43%, S 7.39%.
Properties
White crystalline powder; odorless but has a bitter taste. pH of aq solns about 4.5. Exists in two forms, the usual form mp ≈240°, the other mp ≈198°. Freely sol in water. Practically insol in alcohol, chloroform, ether.Therapeutic Category
Antimalarial; antirheumatic; lupus erythematosus suppressant.
Keywords
Antimalarial; Antiarthritic/Antirheumatic; Lupus Erythematosus Suppressant