4819. 1α-Hydroxycholecalciferol
Nomenclature
Description and references
Synthetic analog of calcitriol, q.v., the hormonal form of vitamin D3, which shows identical potency with respect to stimulation of intestinal calcium absorption and bone mineral mobilization. This common activity is thought to be due either to the presence in both of a 1α hydroxy group or more likely, to the conversion of the 1α-OH-CC in vivo to 1α,25-dihydroxycholecalciferol: Haussler et al., Proc. Natl. Acad. Sci. USA 70, 2248 (1973). Synthesis from cholesterol: Holick et al., Science 180, 190 (1973); Barton et al., J. Am. Chem. Soc. 95, 2748 (1973); Fürst et al., Helv. Chim. Acta 56, 1708 (1973); M. Morisaki et al., Chem. Pharm. Bull. 23, 3272 (1975); T. Sato et al., ibid. 26, 2933 (1979). Total synthesis: R. G. Harrison et al., Tetrahedron Lett. 1973, 3649; eidem, J. Chem. Soc. Perkin Trans. 1 1974, 2654; P. J. Kocienski, B. Lythgoe, ibid. 1980, 1400. Synthesis of the 1β-epimer from the 1α-form and biological properties: H. E. Paaren et al., Chem. Commun. 1977, 890.
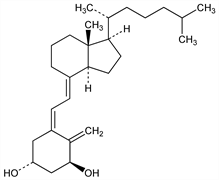
Properties
mp 134-136° (Harrison); also reported as mp 138-139.5° (Fürst). [α]D25 +28° (ether). uv max (ether): 264 nm (ε 18000) (Harrison); also reported as ε 20200 (Barton).Derivative
1β-Epimer.Nomenclature
1β-Hydroxycholecalciferol; 1β-hydroxyvitamin D3.Therapeutic Category
Vitamin D source.
Therapeutic Category (Veterinary)
Vitamin D source.Keywords
Vitamin/Vitamin Source; Vitamin D