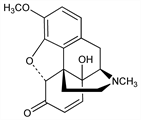
4822. Hydroxycodeinone
Nomenclature
CAS number: 508-54-3
5α-7,8-Didehydro-4,5α-epoxy-14-hydroxy-3-methoxy-17-methylmorphinan-6-one; 14-hydroxycodeinone. C18H19NO4; mol wt 313.35.
C 68.99%, H 6.11%, N 4.47%, O 20.42%.
Description and references
Preparation from thebaine: Freund, Speyer, J. Prakt. Chem. 94, 135 (1916). From codeine: Merck, DE 411530; Frdl. 15, 1516; K. W. Bentley, The Chemistry of the Morphine Alkaloids (Oxford, 1954). Improved synthesis: F. M. Hauser et al., J. Med. Chem. 17, 1117 (1974). Isoln from Papaver bracteatum Lindl.: H. G. Theuns et al., Phytochemistry 16, 753 (1977).
Properties
Plates from 96% alcohol + few drops chloroform, dec 275°. Freely sol in CHCl3, methyl Cellosolve, petr ether, ethyl acetate; slightly sol in alcohol. Practically insol in water, ether; also insol in aq alkaline solns.Derivative
Hydrochloride monohydrate.C18H19NO4.HCl.H2O; mol wt 367.82.
C 58.78%, H 6.03%, N 3.81%, O 21.75%, Cl 9.64%.