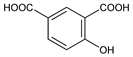
4827. 4-Hydroxyisophthalic Acid
Nomenclature
CAS number: 636-46-4
4-Hydroxy-1,3-benzenedicarboxylic acid. C8H6O5; mol wt 182.13.
C 52.76%, H 3.32%, O 43.92%.
Description and references
Prepd by high pressure carbonation of potassium phenate (Kolbe-Schmitt reaction): Baine et al., J. Org. Chem. 19, 510 (1954) or by boiling salicylic acid with carbon tetrachloride in alkaline medium in the presence of Cu powder: DE 258887 (1913 to Zeltner & Landau); Chem. Zentralbl. 1913, I, 1641; Frdl. 11, 210. Major constituent of the brown dust residue from sublimation of salicylic acid: Hunt et al., Chem. Ind. (London) 1955, 417.