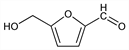
4832. 5-(Hydroxymethyl)-2-furaldehyde
Nomenclature
CAS number: 67-47-0
5-(Hydroxymethyl)-2-furancarboxaldehyde; 5-(hydroxymethyl)-2-furancarbonal; 5-(hydroxymethyl)-2-furfural; HMF; 5-hydroxymethyl-2-formylfuran. C6H6O3; mol wt 126.11.
C 57.14%, H 4.80%, O 38.06%.
Description and references
Prepn from the fructose portion of the sugar molecule in 57% yield: Haworth, Jones, J. Chem. Soc. 1944, 667; Haworth, Wiggins, GB 591858 (1947); GB 600871 (1948). Improved process: Garber, Jones, US 2929823 (1960 to Merck & Co.). Purification: Jones, Lange, US 2994645 (1961 to Merck & Co.). Outline of process using cornstarch, glucose and sucrose as raw materials: Medwick, Chem. Eng. News 75 (Sept. 11, 1961). Prepn from molasses: Jones, Lange, US 3066150 (1962 to Merck & Co.); Hales et al., US 3071599 (1963 to Atlas Chem.).