4839. 17α-Hydroxyprogesterone
Nomenclature
CAS number: 68-96-2
17-Hydroxypregn-4-ene-3,20-dione; 4-pregnen-17α-ol-3,20-dione. C21H30O3; mol wt 330.46.
C 76.33%, H 9.15%, O 14.52%.
Description and references
Isoln from adrenal glands: Pfiffner, North, J. Biol. Chem. 132, 459 (1940); 139, 855 (1941); von Euw, Reichstein, Helv. Chim. Acta 24, 879 (1941). Prepn: Julian et al., US 2648662 (1953 to Glidden); Ringold et al., US 2802839; Stork et al., US 2805203 (both 1957 to Syntex); Chemerda et al., US 2777843; Cutler, Chemerda, US 2786856; US 2786857; Dulaney, McAleer, US 2813060 (all 1957 to Merck & Co.); Cutler et al., J. Org. Chem. 24, 1629 (1959); Pederson, US 3000883 (1961 to Upjohn).
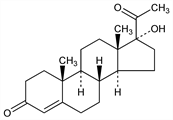
Properties
Rhombic or hexagonal leaflets from acetone or alcohol, mp 222-223° (rapid heating). With slow heating the substance undergoes molecular rearrangement accompanied by partial resolidification and becomes completely molten only at 276°. [α]D17 +105.6° (c = 1.0417 in chloroform).Derivative
Acetate.Nomenclature
17α-Acetoxyprogesterone.C23H32O4; mol wt 372.50.
C 74.16%, H 8.66%, O 17.18%.
Properties
Crystals from chloroform + methanol, mp 239-240°. uv max: 240 nm (log ε 4.33).Derivative
Caproate.Nomenclature
CAS number: 630-56-8
17α-Hydroxyprogesterone hexanoate; Lentogest (Amsa); Proge (Mochida); Proluton Depot (Schering AG). C27H40O4; mol wt 428.60.
C 75.66%, H 9.41%, O 14.93%.
Description and references
Prepn: Kaspar et al., US 2753360 (1956 to Schering AG). Comprehensive description: K. Florey, Anal. Profiles Drug Subs. 4 209-224 (1975).
Properties
Dense needles from isopropyl ether or methanol, mp 119-121°. [α]D20 +61° (c = 1 in chloroform). Soly (mg/ml): sesame oil 25-29; levulinic acid butyl ester 350-400.Therapeutic Category
Progestogen.
Therapeutic Category (Veterinary)
Estrus regulator.Keywords
Progestogen