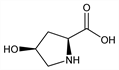
4840. Hydroxyproline
Nomenclature
Description and references
Amino acid analog of proline, q.v.; non-essential for human development. Shows cis-trans isomerism. Constituent of collagen, q.v. Isoln from gelatin: E. Fischer, Ber. 35, 2660 (1902). Synthesis: H. Leuchs, Ber. 38, 1937 (1905); and structure determn: H. Leuchs, J. F. Brewster, Ber. 46, 986 (1913). Stereochemistry: Hudson, Neuberger, J. Org. Chem. 15, 24 (1950). Early chemistry and biochemistry: Amino Acids and Proteins, D. M. Greenberg, Ed. (Charles C. Thomas, Springfield, IL, 1951) 950 pp., passim; J. P. Greenstein, M. Winitz, Chemistry of the Amino Acids vols 1-3 (John Wiley and Sons, Inc., New York, 1961) pp. 2018-2042, passim. Flow sheets of four different syntheses: Chem. Eng. News 40, 40 (Nov. 12, 1962). One pot synthesis: S. G. Ramaswamy, E. Adams, J. Org. Chem. 42, 3440 (1977). Proof of occurence in plants: D. Ashford, A. Neuberger, Trends Biochem. Sci. 5, 245 (1980). Effects of cis-trans isomerism on protein structure: N. Panasik, Jr. et al., Int. J. Pept. Protein Res. 44, 262 (1994). Review: R. Kuttan, A. N. Radhakrishnan, Adv. Enzymol. 37, 273-348 (1973). Review of metabolism: E. Adams, L. Frank, Annu. Rev. Biochem. 49, 1005-1061 (1980).