4866. Hypoglycine A
Nomenclature
CAS number: 156-56-9
(αS,1R)-α-Amino-2-methylenecyclopropanepropanoic
acid; 2-methylenecyclopropanealanine; 2-amino-4,5-methylenehex-5-enoic acid; α-amino-β-(2-methylenecyclopropyl)propionic acid; hypoglycin; hypoglycin A. C7H11NO2; mol wt 141.17.
C 59.56%, H 7.85%, N 9.92%, O 22.67%.
Description and references
Hypoglycemic principle from the akee plant, Blighia sapida Kon., Sapindaceae: Hassall et al., Nature 173, 356 (1954); Hassall, Reyle, Biochem. J. 60, 334 (1955). Structure: de Ropp et al., J. Am. Chem. Soc. 80, 1004 (1958); Renner et al., Helv. Chim. Acta 41, 589 (1958); Ellington et al., J. Chem. Soc. 1959, 80. Synthesis: Carbon et al., J. Am. Chem. Soc. 80, 1002 (1958); Black, Landor, Tetrahedron Lett. 1963, 1065.
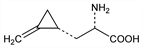
Properties
Yellow plates from methanol + water, mp 280-284°. [α]D32 +9.2°.Derivative
Methyl ester hydrochloride.C8H13NO2.HCl; mol wt 191.66.
C 50.13%, H 7.36%, N 7.31%, O 16.70%, Cl 18.50%.
Properties
Needles from methanol + ether, mp 151-152°. [α]D22 +36° (c = 2.0).Derivative
Hypoglycine B.Nomenclature
CAS number: 502-37-4
N-l-γ-Glutamyl-3-(2-methylenecyclopropyl)alanine; γ-l-glutamylhypoglycine. C12H18N2O5; mol wt 270.28.
C 53.33%, H 6.71%, N 10.36%, O 29.60%.
Description and references
Structure: Jhl, Stoll, Helv. Chim. Acta 42, 156 (1959); Hassall, John, J. Chem. Soc. 1960, 4112. Synthesis: Jhl, Stoll, Helv. Chim. Acta 42, 716 (1959).