4891. Idoxuridine
Nomenclature
CAS number: 54-42-2
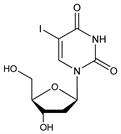
2′-Deoxy-5-iodouridine; 1-(2-deoxy-β-d-ribofuranosyl)-5-iodouracil; 5-iodo-2′-deoxyuridine; IdU; IdUR; IUdR; IdUrd; Dendrid (Alcon); Emanil; Herpes-Gel (Master); Herplex (Allergan); Idexur; Idoxene (Spodefell); Idulea; Iduridin (Ferring); Kerecid; Ophthalmadine (SAS); Stoxil (SK & F); Virudox (Bioglan). C9H11IN2O5; mol wt 354.10.
C 30.53%, H 3.13%, I 35.84%, N 7.91%, O 22.59%.
Description and references
Cytotoxic nucleoside with antiviral and antineoplastic activity. Prepn: Prusoff, Biochim. Biophys. Acta 32, 295 (1959); Cheong et al., J. Biol. Chem. 235, 1441 (1960); Chang, Welch, J. Med. Chem. 6, 428 (1963); Amiard, Torelli, FR 1336866 (1963 to Roussel-UCLAF), C.A. 60, 3082g (1964); GB 1024156; Prystas, Sorm, Collect. Czech. Chem. Commun. 29, 121 (1964). Crystal and molecular structure: Camerman, Trotter, Acta Crystallogr. 18, 203 (1965). Review: W. H. Prusoff et al. in Antibiotics vol. 5 (pt. 2), F. E. Hahn, Ed. (Springer-Verlag, New York, 1979) pp 236-261.
Properties
Crystals from water, triclinic, dec 160° (Prusoff; Chang, Welch), 190-195° (Cheong et al.), 240° (Amiard, Torelli), over 175° (Prystas, Sorm). uv max (water): 288 nm (log ε 3.87). [α]D25 +7.4° (c = 0.108 in water); [α]D20 +29° (N soda). Physical properties: Ravin, Gulesich, J. Am. Pharm. Assoc. [NS] 4, 122 (1964). pKa 8.25. pH of 0.1% aq soln, about 6. Soly at 25° in mg/ml: 2.0 in water; 2.0 in 0.2N HCl; 74.0 in 0.2N NaOH; 4.4 in methanol; 2.6 in alc; 0.014 in ether; 0.003 in chloroform; 1.6 in acetone; 1.8 in ethyl acetate; 5.7 in dioxane. LD50 i.p. in mice: 2.5 g/kg (Prusoff, 1979).Derivative
α-Anomer.Nomenclature
1-(2-Deoxy-α-d-erythro-pentofuranosyl)-5-iodouracil; 1-(2-deoxy-α-d-ribofuranosyl)-5-iodouracil; α-2′-deoxy-5-iodouridine.Properties
Crystals from water, dec 170°. [α]D25 +21.8° (c = 0.170). uv max (water): 288 nm (log ε 3.88).Therapeutic Category
Antiviral.
Keywords
Antiviral; Nucleosides/Nucleotides