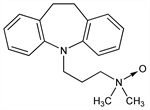
4921. Imipramine N-Oxide
Nomenclature
Description and references
Identification as a metabolite of imipramine, q.v.: V. Fishman, H. Goldenberg, Proc. Soc. Exp. Biol. Med. 110, 187 (1962). Prepn: FR M2508; H. Dyrsting, J. B. Pedersen, US 3574852 (1964, 1971, both to Dumex). Pharmacology: W. Theobald et al., Med. Pharmacol. Exp. 15, 187 (1966). Metabolism: M. H. Bickel, M. Baggiolini, Biochem. Pharmacol. 15, 1155 (1966); R. Minder et al., Arch. Pharmakol. 268, 334 (1971). GLC determn in biological samples: H. J. Weder, M. H. Bickel, J. Chromatogr. 37, 181 (1968); HPLC determn: E. Koyama et al., Ther. Drug Monit. 15, 224 (1993). Clinical trial: W. Rapp et al., Acta Psychiatr. Scand. 49, 77 (1973). Clinical pharmacokinetics: A. Nagy, T. Hansen, Acta Pharmacol. Toxicol. 42, 58 (1978).
Properties
White needle-shaped crystals, mp 120-123° (dec). Sol in methanol, ether, acetone and benzene. Strongly hygroscopic.Derivative
Monohydrate.Properties
mp 75-79°.Derivative
Hydrochloride.Nomenclature
Properties
Colorless crystalline powder, mp 167-174°; also reported as mp 153-155° (dec). Very bitter taste. An aq. soln is moderately acidic. Sol in 75 parts water, in 12 parts 95% ethanol, 4.5 parts chloroform. Nearly insol in ether. LD50 in rats, mice (mg/kg): 90, 150 i.p. (Dyrsting, Pedersen).Therapeutic Category
Antidepressant.
Keywords
Antidepressant; Tricyclics