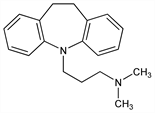
4920. Imipramine
Nomenclature
CAS number: 50-49-7
10,11-Dihydro-N,N-dimethyl-5H-dibenz[b,f]azepine-5-propanamine; 5-(3-dimethylaminopropyl)-10,11-dihydro-5H-dibenz[b,f]azepine; N-(γ-dimethylaminopropyl)iminodibenzyl; imizin; G-22355. C19H24N2; mol wt 280.41.
C 81.38%, H 8.63%, N 9.99%.
Description and references
Tricyclic antidepressant. Prepn: Haefliger, Schindler, US 2554736 (1951 to Geigy); eidem, Helv. Chim. Acta 37, 472 (1954). Reviews of pharmacology: Crismon, Psychopharmacol. Bull. 4, 151 pp (Oct. 1967); Glassman, Perel, Arch. Gen. Psychiatry 28, 649 (1973). Comprehensive description: D. N. Kender, R. E. Schiesswohl, Anal. Profiles Drug Subs. 14, 37-75 (1985). Comparative clinical trials in depression: R. S. Lipman et al., Arch. Gen. Psychiatry 43, 68 (1986); in anxiety: R.J. Kahn et al., ibid. 79. Clinical trial for panic disorder: D. H. Barlow et al., J. Am. Med. Assoc. 283, 2529 (2000).
Properties
Free base, bp0.1 160°.Derivative
Hydrochloride.Nomenclature
CAS number: 113-52-0
Chrytemin (Fujinaga); Imidol (Mitsubishi); Melipramine (EGIS); Pryleugan (Temmler); Tofranil (Mallinckrodt). C19H24N2.HCl; mol wt 316.87.
C 72.02%, H 7.95%, N 8.84%, Cl 11.19%.
Properties
Crystals from acetone, mp 174-175°. Acquires a yellow to reddish discoloration under the influence of light. Freely sol in water, less sol in alcohol, sparingly sol in acetone. LD50 in mice, rats (mg/kg): 400, 490 orally; 110, 90 i.p. See: A. Tobe et al., Arzneim.-Forsch. 31, 1278 (1981).Derivative
Pamoate.Nomenclature
CAS number: 10075-24-8
Tofranil-PM (Mallinckrodt). (C19H24N2)2.C23H16O6; mol wt 949.18.
C 77.19%, H 6.80%, N 5.90%, O 10.11%.
Properties
Fine yellow, tasteles, odorless powder. Sol in ethanol, acetone, ether, chloroform, carbon tetrachloride. Insol in water.Therapeutic Category
Antidepressant.
Therapeutic Category (Veterinary)
In treatment of cataplexy, urinary incontinence, narcolepsy, ejaculatory dysfunction, and behavioral disorders.Keywords
Antidepressant; Tricyclics