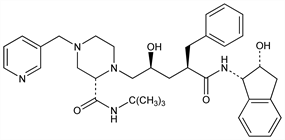
4945. Indinavir
Nomenclature
Description and references
Member of the novel hydroxyaminopentane amide class of HIV-1 protease inhibitors. Prepn: J. P. Vacca et al., EP 541168; eidem, US 5413999 (1993, 1995 both to Merck & Co.); B. D. Dorsey et al., J. Med. Chem. 37, 3443 (1994). Diastereoselective synthesis: D. Askin et al., Tetrahedron Lett. 35, 673 (1994); P. E. Maligres et al., ibid. 36, 2195 (1995). Crystal structure of HIV-II protease complex: Z. Chen et al., J. Biol. Chem. 269, 26344 (1994). Antiviral activity and pharmacokinetics: J. P. Vacca et al., Proc. Natl. Acad. Sci. USA 91, 4096 (1994). HPLC determn in plasma and urine: E. Woolf et al., J. Chromatogr. A 692, 45 (1995). Metabolism in humans: S. K. Balani et al., Drug Metab. Dispos. 23, 266 (1995). Review of clinical pharmacology and therapeutic efficacy: G. L. Plosker, S. Noble, Drugs 58, 1165-1203 (1999).
Properties
Occurs as monohydrate, crystals from wet ethyl or isopropyl acetate, loss of water below 100° followed by recrystallization of anhydrous form with mp 153-154°; second anhydrous form, mp 167.5-168°. Soly in water (mg/ml): 0.015 (unbuffered), >1.5 (pH 4.0). [α]D22 +24.1° (c = 0.0133 in chloroform).Derivative
Sulfate.Nomenclature
Properties
White to off-white, hygroscopic, crystalline powder. Occurs as the monoethanolate, crystals from absolute ethanol, softens at 135°, mp 150-153° (dec). Converts to hydrate upon loss of ethanol and exposure to moist air. Sol in water and methanol.Therapeutic Category
Antiretroviral.
Keywords
HIV Protease Inhibitor; Antiretroviral; Protease Inhibitors