4975. Inosine
Nomenclature
CAS number: 58-63-9
Hypoxanthine riboside; 9-β-d-ribofuranosylhypoxanthine; hypoxanthosine; Inosie (Morishita); Oxiamine (Made); Ribonosine (Toyo Jozo); Trophicardyl. C10H12N4O5; mol wt 268.23.
C 44.78%, H 4.51%, N 20.89%, O 29.82%.
Description and references
In meat and meat extracts, in sugar beets. Prepd from adenosine by incubation with purified adenosine deaminase from intestine: Kalckar, J. Biol. Chem. 167, 445 (1947); also by the action of sodium nitrite and acetic acid on adenosine: Levene, Jacobs, Ber. 43, 3161 (1910); by the use of barium nitrite and H2SO4: Reiff et al., US 3049536 (1962 to Zellstoff-Fabrik Waldhof). Fermentation method: Motozaki et al., US 3111459 (1963 to Ajinomoto). Structure: Bredereck, Ber. 66, 198 (1933); Z. Physiol. Chem. 223, 61 (1934); Gulland, Holiday, J. Chem. Soc. 1936, 765.
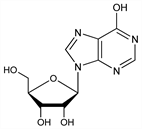
Derivative
Dihydrate.Properties
Long rectangular plates from water, mp 90°. Anhydrous needles from 80% alc, dec 218° (rapid heating). [α]D18 49.2° (c = 0.9 in H2O). [α]20white 73° (0.5 g + 2 ml N NaOH + 3 ml H2O). 100 ml of the satd water soln at 20° contain 1.6 g inosine. Absorption spectrum: Kalckar, loc. cit. uv max (pH 6.0): 248.5 nm (ε 12200). Boiling with 0.1N H2SO4 yields hypoxanthin and d-ribose.Therapeutic Category
Activates cellular functions.