4977. Inosinic Acid
Nomenclature
CAS number: 131-99-7
5′-Inosinic acid; 5-inosinic acid; muscle inosinic acid; t-inosinic acid; hypoxanthine riboside-5-phosphoric acid; IMP. C10H13N4O8P; mol wt 348.21.
C 34.49%, H 3.76%, N 16.09%, O 36.76%, P 8.90%.
Description and references
Prepn from meat extract: Levene, Bass, Nucleic Acids (New York, 1931) p 229; from dried sardines: Yoshida, Kageyama, JP 56 732 (1956 to Ajinomoto), C.A. 51, 3870b (1957). Structure: Levene, Bass, op. cit., pp 187-192; Bredereck, Ber. 66, 198 (1933); Levene, Tipson, J. Biol. Chem. 111, 313 (1935). Also prepd from muscle by enzymatic deamination of muscle adenylic acid: Ostern, Biochem. Z. 254, 65 (1932); by hydrolysis of inosine triphosphate: Kleinzeller, Biochem. J. 36, 729 (1942). Studies on the enzymatic synthesis: Greenberg, J. Biol. Chem. 190, 611 (1951); Korn et al., ibid. 217, 875 (1955). Microbial fermentation method using mutant strains of Micrococcus glutamicus: Kinoshita et al., US 3232844 (1966 to Kyowa).
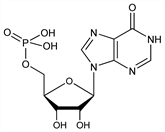
Properties
Syrup, solidifies to a glass when dried over H2SO4. Agreeable sour taste. pK1 = 2.4; pK2 = 6.4. Absorption spectrum: Kalckar, J. Biol. Chem. 167, 445 (1947). Freely sol in water, in formic acid; very sparingly sol in alcohol, ether. On boiling with acid hydrolyzes to 1 mol H3PO4, 1 mol hypoxanthine, 1 mol d-ribose.Derivative
Disodium salt dihydrate.C10H11N4Na2O8P.2H2O; mol wt 428.20.
C 28.05%, H 3.53%, N 13.08%, Na 10.74%, O 37.36%, P 7.23%.
Properties
Barely sol in alcohol, ether, acetone; soly in water at 20° about 13 g/100 ml. Kawasaki, New Food Ind. 3, no. 1, 17 (1961).Derivative
Barium salt.C10H11BaN4O8P; mol wt 483.52.
C 24.84%, H 2.29%, Ba 28.40%, N 11.59%, O 26.47%, P 6.41%.