5022. Iodipamide
Nomenclature
CAS number: 606-17-7
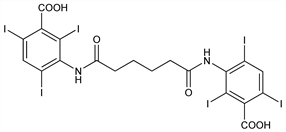
3,3′-[(1,6-Dioxo-1,6-hexanediyl)diimino]bis[2,4,6-triiodobenzoic
acid]; 3,3′-(adipoyldiimino)bis[2,4,6-triiodobenzoic acid]; N,N′-adipylbis(3-amino-2,4,6-triiodobenzoic acid); adipic acid di(3-carboxy-2,4,6-triiodoanilide); adipiodone; Cholografin (BMS); Cholospect. C20H14I6N2O6; mol wt 1139.76.
C 21.08%, H 1.24%, I 66.81%, N 2.46%, O 8.42%.
Description and references
Prepn: Priewe, Rutkowski, US 2776241 (1957 to Schering AG); Kotler-Brajtburg et al., Rocz. Chem. 36, 763 (1962), C.A. 58, 5568a (1963). Purification: Cassebaum, Drux, DD 33738 (1964), C.A. 63, 11441b (1965). Pharmacology: E. Fischer, F. Varga, Acta Physiol. Acad. Sci. Hung. 38, 135 (1970). Metabolic studies: Kiyono et al., Radioisotopes 20, 78 (1971). Pharmacology and toxicity study: F. J. Rosenberg et al., Invest. Radiol. 15, S142 (1980). Comprehensive description: H. H. Lerner, Anal. Profiles Drug Subs. 3, 333-363 (1974).
Properties
Crystals, dec 306-308°. nD21.5 1.3294 (c = 0.445 in methanol). Soly at 20°: methanol 0.8%; ethanol 0.3%; acetone 0.2%; ether 0.1%. Practically insol in water, benzene. LD50 in mice, rats (mg/kg): 2380 ±290, 4430 ±310 i.v. (Rosenberg).Derivative
Disodium salt.Nomenclature
CAS number: 2618-26-0
C20H12I6N2Na2O6; mol wt 1183.73.
C 20.29%, H 1.02%, I 64.32%, N 2.37%, Na 3.88%, O 8.11%.
Properties
Prepd by dissolving the free acid in a dil aq soln of NaOH and buffering to pH 6.5- 7.7. Used as 20% soln.Derivative
Bis[N-methylglucamine] salt.Nomenclature
CAS number: 3521-84-4
Iodipamide meglumine; Biligrafin (Schering AG); Endocistobil (Bracco); Endografin (Schering AG); Intrabilix; Transbilix (Guerbet). C34H48I6N4O16; mol wt 1530.19.
C 26.69%, H 3.16%, I 49.76%, N 3.66%, O 16.73%.
Properties
More sol in water than the disodium salt. Used as 40% soln.Therapeutic Category
Diagnostic aid (radiopaque medium—cholecystographic).
Therapeutic Category (Veterinary)
Diagnostic aid (radiopaque medium).Keywords
Diagnostic Aid (Radiopaque Medium)