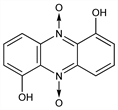
5021. Iodinin
Nomenclature
CAS number: 68-81-5
1,6-Phenazinediol 5,10-dioxide; 1,5-dihydroxyphenazine N,N′-dioxide. C12H8N2O4; mol wt 244.20.
C 59.02%, H 3.30%, N 11.47%, O 26.21%.
Description and references
Antibiotic pigment from Chromobacterium iodinum: Clemo, McIlwain, J. Chem. Soc. 1938, 479; Hegedüs in Emil Barell Jubilee Volume (1946), p 388; from Waksmania aerata and Pseudomonas iodinum: Gerber, Lechevalier, Biochemistry 3, 598 (1964). Structure: Clemo, Daglish, J. Chem. Soc. 1950, 1481. Synthesis: Matsumura, Takeda, Nippon Kagaku Zasshi 81, 515 (1960), C.A. 56, 470a (1962). Biosynthetic studies on incorporation of shikimic acid, q.v., into iodinin: U. Hollstein et al., Tetrahedron Lett. 1978, 2987; R. B. Herbert et al., J. Chem. Soc. Perkin Trans. 1 1979, 2411; T. Etherington et al., ibid. 2416.