5038. Iodophthalein Sodium
Nomenclature
CAS number: 2217-44-9
Soluble iodophthalein; tetraiodophenolphthalein sodium; T.I.P.P.S.; tetraiodophthalein sodium; tetiothalein sodium; Iodeikon (Mallinckrodt); Cholepulvis; Keraphen; Shadocol; Bilitrast; Iodognost; Stipolac (Burroughs Wellcome); Tetraiode; Foriod; Iodtetragnost; Antinosin; Cholumbrin; Iodorayoral; Opacin; Photobiline; Piliophen; Videophel; Radiotetrane; Nosophene sodium; Iodophene sodium. C20H8I4Na2O4; mol wt 865.87.
C 27.74%, H 0.93%, I 58.63%, Na 5.31%, O 7.39%.
Description and references
Isomeric with phentetiothalein sodium, q.v. Prepd by the action of iodine (in KI soln) on phenolphthalein in aq alk soln: Classen, Lb, Ber. 28, 1603 (1895); DE 85930 (1894); DE 88390 (1895). Also obtained by treating an alk soln of phenolphthalein with iodine monochloride: Kalle, DE 143596 (1900). Prepn of free acid: Orndorff, Mahood, J. Am. Chem. Soc. 40, 937 (1918). Toxicity data: J. O. Hoppe et al., J. Med. Chem. 13, 997 (1970). Metabolism in rabbits: D. Pitre, L. Fumagalli, Farmaco Ed. Sci. 32, 76 (1977).
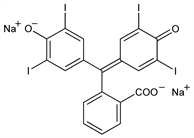
Derivative
Trihydrate.Properties
Pale-blue to violet crystals. Saline and astringent taste. Somewhat hygroscopic. Gradually decomp on exposure to air due to absorption of CO2 and becomes incompletely sol. One g dissolves in about 7 ml water, forming a clear, deep-blue dichroic soln. Slightly sol in alc. Keep well closed. LD50 in mice (mg/kg): 360 i.v., 3800 orally (Hoppe).Derivative
Free acid.Nomenclature
CAS number: 386-17-4
Iodophthalein; tetraiodophthalein; Iodophene; Nosophen. C20H10I4O4; mol wt 821.91.
C 29.23%, H 1.23%, I 61.76%, O 7.79%.
Properties
Light-yellow, odorless, tasteless powder; dec at about 200°. Practically insol in water. Slightly sol in alcohol; sol in alkalies, chloroform, ether.Therapeutic Category
Diagnostic aid (radiopaque medium—cholecystographic).
Keywords
Diagnostic Aid (Radiopaque Medium)