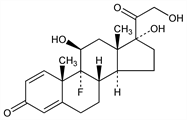
5174. Isoflupredone
Nomenclature
Description and references
Prepn: J. Fried et al., J. Am. Chem. Soc. 77, 4181 (1955). Microbiological prepn: A. Nobile et al., J. Am. Chem. Soc. 77, 4184 (1955); E. Vischer et al., Helv. Chim. Acta 38, 1502 (1955); T. H. Stoudt et al., Arch. Biochem. Biophys. 59, 304 (1955). Prepn of the 21-acetate: R. F. Hirschmann et al., ibid. 3166; J. A. Hogg et al., ibid. 4438; Ch. Meystre et al., Helv. Chim. Acta 39, 734 (1956); A. Wellstein et al., DE 1020329 (1957 to Ciba), C.A. 54, 25562h (1960); GB 843214 (1960 to Schering), C.A. 55, 5597b (1961). Absorption spectra: L. L. Smith, W. H. Muller, J. Org. Chem. 23, 960 (1958). Mass spectrometry: P. Toft et al., Can. J. Pharm. Sci. 7, 53 (1972). Urinary excretion in horses: D. I. Chapman et al., Vet. Rec. 100, 447 (1977).