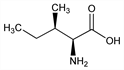
5179. Isoleucine
Nomenclature
Description and references
An essential amino acid for human development. Isoln from beet-sugar mother liquors: F. Ehrlich, Ber. 37, 1809 (1904). Structure and identification of isomers: idem, Ber. 40, 2538 (1907); J. P. Greenstein et al., J. Biol. Chem. 204, 307 (1953). Early chemistry and biochemistry: Amino Acids and Proteins, D. M. Greenberg, Ed. (Charles C. Thomas, Springfield, IL, 1951) 950 pp., passim; J. P. Greenstein, M. Winitz, Chemistry of the Amino Acids vol 1-3 (John Wiley and Sons, Inc., New York, 1961) pp. 2043-2074, passim. TLC determ in serum from patients with maple sugar urine disease (MSDU): R. J. Allen et al., Clin. Chem. 18, 413 (1972). Metabolism of Ile and isomers in MSDU: U. Wendel et al., Pediatr. Res. 25, 11 (1989); and clinical consequences: T. Mogos et al., Rev. Roum. Med. Interne 32, 57 (1994). Review of biosynthesis: M. Iaccarino et al., Curr. Top. Cell. Regul. 14, 29-73 (1978).