5186. Isoniazid
Nomenclature
Description and references
Prepn: Meyer, Mally, Monatsh. Chem. 33, 400 (1912); Lock, Pharm. Ind. 14, 366 (1952); Urbanski et al., Rocz. Chem. 27, 161 (1953); Gasson, US 2830994 (1958 to Distillers). Compositions for combating tuberculosis: H. H. Fox, US 2596069 (1952 to Hoffmann-La Roche). Pharmacokinetics: W. W. Weber, D. W. Hein, Clin. Pharmacokinet. 4, 401 (1979). Mechanism of action study: K. Johnsson et al., J. Am. Chem. Soc. 117, 5009 (1995). Toxicity data: E. H. Jenney, C. C. Pfeiffer, J. Pharmacol. Exp. Ther. 122, 110 (1958). Evaluation of carcinogenic risk: IARC Monographs 4, 159 (1974). Comprehensive description: G. A. Brewer, Anal. Profiles Drug Subs. 6, 183-258 (1977). Clinical trial in HIV-positive patients: J. W. Pape et al., Lancet 342, 268 (1993). Review of use in prevention and treatment of TB: American Thoracic Society, Am. Rev. Respir. Dis. 134, 355-363 (1986); T. J. Jordan et al., ibid. 144, 1357-1360 (1991).
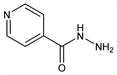
Properties
Crystals from alc, mp 171.4°. uv max (water): 266 nm (E1%1cm 378); (0.01N HCl): 265 nm (E1%1cm ≈420). Soly in water at 25°: about 14%; at 40°: about 26%; in alc at 25°: about 2%, in boiling alc: about 10%; in chloroform: about 0.1%. Practically insol in ether, benzene. pH of a 1% aq soln 5.5 to 6.5. Aqueous solns may be sterilized at 120° for 30 min. LD50 in mice (mg/kg): 151 i.p., 149 i.v. (Jenney, Pfeiffer).Derivative
4-Aminosalicylate.Nomenclature
Description and references
An equimolecular salt produced by dissolving the components in hot dil alc, followed by cooling and evapn: Charonnat, Boime, Compt. Rend. 236, 2140 (1953). Prepn: CH 303085 (1955 to Hoffmann-La Roche).
Properties
Yellow crystals from methanol or ethanol, mp 142-144°. Sparingly sol in water. uv max 272, 303 nm (E1%1cm 550, 445).Derivative
Methanesulfonate.Nomenclature
Description and references
Prepn: Logemann, US 2759944 (1956 to Carlo Erba).
Properties
Crystals, dec 187-189°.Derivative
Methanesulfonate sodium.Nomenclature
Properties
Yellow crystals from water, dec 164-167°.Therapeutic Category
Antibacterial (tuberculostatic).
Therapeutic Category (Veterinary)
Antibacterial (tuberculostatic); anti-actinomycotic agent.Keywords
Antibacterial (Tuberculostatic)