5187. Isonicotinic Acid
Nomenclature
CAS number: 55-22-1
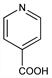
4-Pyridinecarboxylic acid; γ-pyridinecarboxylic acid; γ-picolinic acid. C6H5NO2; mol wt 123.11.
C 58.54%, H 4.09%, N 11.38%, O 25.99%.
Description and references
Prepd by permanganate oxidation of γ-picoline: Gilman, Broadbent, J. Am. Chem. Soc. 70, 2757 (1948); Fields, US 2946801 (1960 to Standard Oil); from 4-ethylpyridine: Wibaut, Arens, Rec. Trav. Chim. 60, 137 (1941); from citric acid to citrazinic acid to 2,6-dichloroisonicotinic acid: Behrmann, Hofmann, Ber. 17, 2681 (1884); Wibaut, Rec. Trav. Chim. 63, 141 (1944). Prepn of ethyl ester: Burrus, Powell, J. Am. Chem. Soc. 67, 1469 (1945).
Properties
Platelets, mp 319°. Sublimes at 260° and 15 mm pressure. pK (25°) 4.96. pH of satd aq soln 3.6. Sparingly sol in cold water (0.52 g/100 ml at 20°), more sol in hot water. Practically insol in benzene, ether, boiling alcohol.Derivative
Methyl ester.C7H7NO2; mol wt 137.14.
C 61.31%, H 5.14%, N 10.21%, O 23.33%.