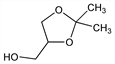
5213. Isopropylidene Glycerol
Nomenclature
CAS number: 100-79-8
2,2-Dimethyl-1,3-dioxolane-4-methanol; 2,2-dimethyl-4-hydroxymethyl-1,3-dioxolane; glycerol dimethylketal; acetone glycerol; Solketal. C6H12O3; mol wt 132.16.
C 54.53%, H 9.15%, O 36.32%.
Description and references
Prepn: Fisher, Ber. 28, 1167 (1895); Fisher, Pfhler, Ber. 53, 1607 (1920); Hibbert, Morazain, Can. J. Res. 2, 38 (1930); Smith, Lindberg, Ber. 64, 510 (1931); M. M. Maglio, C. A. Burger, J. Am. Chem. Soc. 68, 529 (1946); M. Renoll, M. S. Newman, Org. Synth. coll. vol. III, 502 (1955); Mikschik, AT 180926 (1955 to Chemomedica Chemikalien), C.A. 49, 15951 (1955); Williams, GB 802022 (1958 to Peter Spence & Sons); GB 819835 (1959 to Bayer); Perez et al., ES 499129 (1982 to Calipe), C.A. 97, 55794v (1982). Synthesis of d(+)-form: E. Baer, H. Fischer, J. Biol. Chem. 128, 463 (1939); of l()-form: eidem, J. Am. Chem. Soc. 61, 761 (1939). Conversion of d(+)- to l()-form: eidem, ibid. 67, 944 (1945).