Nomenclature
CAS number: 108-20-3
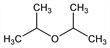
2,2′-Oxybispropane; 2-isopropoxypropane; diisopropyl ether; DIPE.
C
6H
14O; mol wt 102.17.
C 70.53%, H 13.81%, O 15.66%.
Description and references
Prepn: E. Erlenmeyer, Ann. 126, 305 (1863). Physical properties
and solubility: H. R. Fife, E. W. Reid, Ind.
Eng. Chem. 22, 513 (1930). Manuf process:
M. N. Harandi, H. Owen, US 5208387 (1993 to Mobil). GC determn in gasolines: N.
G. Johansen, J. High Resolut. Chromatogr.
Chromatogr. Commun. 7, 487 (1984). Acute toxicity:
E. T. Kimura et al., Toxicol.
Appl. Pharmacol. 19, 699 (1971). Comparison
with MTBE, ETBE and TAME, q.q.v., as gasoline oxygenate: M. J. McNally et al., Oil Gas J. 90, 39 (May 25, 1992).
Properties
Liquid. bp760 68.27°. fp 85.89°. d420 0.72813. nD20 1.36888. Surface tension in air (23°): 32 dynes/cm.
Viscosity (25°): 0.379 cP. Vapor pressure (20°): 158 mm Hg. Specific
heat (22-27°): 0.526 cal/g. Flash pt, closed cup: 6.7°F. Latent heat of vaporization (67.5°): 68.2 cal/g. Soly
of commercial grade in water (19°): 1.71 wt%. Misc with most organic
solvents. Ethanolamines, glycols, glycerol and their monoesters have
limited soly; diesters are sol. Readily forms peroxides when exposed
to oxygen unless stabilized with inhibitors such as hydroquinone or
BHT. LD50 in 14 day old, young adult,
adult rats (ml/kg): 6.4, 16.5, 16.0 orally (Kimura).Caution
Potential symptoms of overexposure
are irritation of eyes, skin and nose; respiratory discomfort; dermatitis.
See NIOSH Pocket Guide to Chemical
Hazards (DHHS/NIOSH 97-140, 1997) p 182.Use
As organic solvent; fuel additive.
