5219. Isopyrocalciferol
Nomenclature
CAS number: 474-70-4
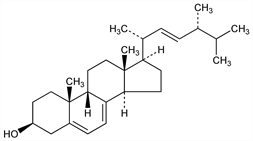
(3β,9β,22E)-9-Ergosta-5,7,22-trien-3-ol; 9β-ergosterol. C28H44O; mol wt 396.65.
C 84.78%, H 11.18%, O 4.03%.
Description and references
Differs sterically from pyrocalciferol at C-10, from ergosterol at C-9. See ref under Pyrocalciferol. Stereochemistry: Castells et al., J. Chem. Soc. 1959, 1159. Selenium dehydrogenation gives Diel's hydrocarbon (3′-methyl-1,2-cylopentenophenanthrene).
Properties
Prisms from ether-methanol. mp 112-115°. uv max: 262, 280 nm. [α]D20 +332°, [α]20546 +415° (c = 1.5 in chloroform). Precipitated by digitonin.Derivative
Acetate.C30H46O2; mol wt 438.69.
C 82.14%, H 10.57%, O 7.29%.