7997. Pyrocalciferol
Nomenclature
CAS number: 128-27-8
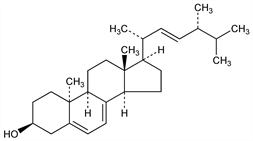
(3β,10α,22E)-Ergosta-5,7,22-trien-3-ol; 9α-lumisterol; 9α-lumista-5,7,22-trien-3β-ol. C28H44O; mol wt 396.65.
C 84.78%, H 11.18%, O 4.03%.
Description and references
Differs from ergosterol in the steric configuration at C-9 and C-10. Formation via isopyrocalciferol: Askew et al., Proc. R. Soc. London B109, 488 (1932); Windaus et al., Nachr. Ges. Wiss. Goettingen Math.-Phys. Kl. 1932, 150. Dehydrogenation with selenium gives Diel's hydrocarbon (3′-methyl-1,2-cyclopentenophenanthrene). Fieser, Fieser, Steroids (New York, 1959) pp 136-143, argue the configuration at C-9 and C-10. See also Castells et al., J. Chem. Soc. 1959, 1159. Critical examination and comparison with other unnatural steroids: Castells et al., ibid. 1962, 2907.
Properties
Needles from methanol, mp 93-95°. [α]D20 +512°, [α]20546 +624° (c = 0.15 in alcohol). uv max: 274, 294 nm (Askew).Derivative
Acetate.C30H46O2; mol wt 438.69.
C 82.14%, H 10.57%, O 7.29%.
Properties
mp 81-82°, [α]D +403° (chloroform).Derivative
3,5-Dinitrobenzoate.C35H46N2O6; mol wt 590.75.
C 71.16%, H 7.85%, N 4.74%, O 16.25%.