5238. Isoxsuprine
Nomenclature
CAS number: 395-28-8
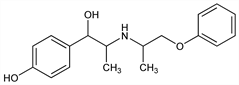
4-Hydroxy-α-[1-[(1-methyl-2-phenoxyethyl)amino]ethyl]benzenemethanol; p-hydroxy-α-[1-[(1-methyl-2-phenoxyethyl)amino]ethyl]benzyl
alcohol; p-hydroxy-N-(1-methyl-2-phenoxyethyl)norephedrine; 1-(p-hydroxyphenyl)-2-(1-methyl-2-phenoxyethylamino)-1-propanol; 2-(3-phenoxy-2-propylamino)-1-(p-hydroxyphenyl)-1-propanol. C18H23NO3; mol wt 301.38.
C 71.73%, H 7.69%, N 4.65%, O 15.93%.
Description and references
Prepn: Moed, van Dijk, Rec. Trav. Chim. 75, 1215 (1956); GB 832286 and GB 832287 (both 1960 to N. V. Philip Gloeilampenfabrieken); Moed, US 3056836 (1962 to N. Am. Philips). Configuration: Dirkx, Rec. Trav. Chim. 83, 535 (1964). Toxicity data: Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971). Clinical evaluation in Raynaud's phenomenon: H. Wesseling et al., Eur. J. Clin. Pharmacol. 20, 329 (1981); in intermittent claudication: S. H. Skotnicki et al., Angiology 35, 685 (1984). Veterinary trial in navicular disease: A. S. Turner, C. M. Tucker, Equine Vet. J. 21, 338 (1989).
Properties
Crystals, mp 102.5-103.5°.Derivative
Hydrochloride.Nomenclature
CAS number: 579-56-6
Dilavase (Organon); Duvadilan (Duphar); Duviculine (Duphar); Navilox (Univet); Suprilent (Duphar); Vadosilan (BMS); Vasodilan (BMS); Vasotran (BMS). C18H23NO3.HCl; mol wt 337.84.
C 63.99%, H 7.16%, N 4.15%, O 14.21%, Cl 10.49%.
Properties
Bitter crystals from water, mp 203-204°. Soly in water at 25° about 2.0% w/v; also sol in ethanol. LD50 in rats (mg/kg): 1750 orally; 164 i.p. (Goldenthal).Therapeutic Category
Vasodilator (peripheral).
Therapeutic Category (Veterinary)
In treatment of navicular disease.Keywords
Vasodilator (Peripheral)