5240. Isradipine
Nomenclature
Description and references
Dihydropyridine calcium channel blocker. Prepn: P. Neumann, DE 2949491; idem, US 4466972 (1980, 1984 both to Sandoz). Prepn of enantiomers: A. Vogel, DE 3320616 (1983 to Sandoz), C.A. 101, 7162s (1984). Comparative study of in vitro effects on human and canine cerebral arteries: E. Müller-Schweinitzer, P. Neumann, J. Cereb. Blood Flow Metab. 3, 354 (1983). Effect on α-adrenoceptor mediated vasoconstriction in rats: K. Jie et al., Arch. Int. Pharmacodyn. 278, 72 (1985). Pharmacokinetics: F. L. S. Tee, J. M. Jaffe, Eur. J. Clin. Pharmacol. 32, 361 (1987). Clinical evaluation in angina and coronary artery disease: C. E. Handler, E. Sowton, ibid. 27, 415 (1984); in hypertension: E. B. Nelson et al., Clin. Pharmacol. Ther. 40, 694 (1986). Comparison of hemodynamic effects of enantiomers: R. P. Hof et al., J. Cardiovasc. Pharmacol. 8, 221 (1986). Series of articles on pharmacology and clinical use: Am. J. Med. 86, 1-146 (1989).
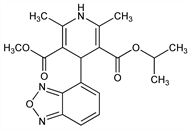
Properties
mp 168-170°.Derivative
S(+)-Form.Nomenclature
PN-205-033.Properties
Crystals from ether + hexane, mp 142°. [α]D20 +6.7° (c = 1.5 in ethanol).Derivative
R()-Form.Nomenclature
PN-205-034.Properties
Crystals from ether + hexane, mp 140°. [α]D20 6.7° (c = 1.67 in ethanol).Therapeutic Category
Antihypertensive; antianginal.
Keywords
Antianginal; Antihypertensive; Dihydropyridine Derivatives; Calcium Channel Blocker; Dihydropyridine Derivatives