5258. Jasmolins
Description and references
Active insecticidal constituents of pyrethrum flowers. Isoln and structure: Godin et al., J. Econ. Entomol. 58, 548 (1965); Godin et al., J. Chem. Soc. C 1966, 322. Stereochemistry: Begley et al., Chem. Commun. 1972, 1276. Review of toxicology and human exposure: Toxicological Profile for Pyrethrins and Pyrethroids (PB2004-100004, 2003) 332 pp.
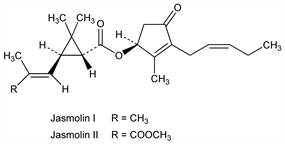
Derivative
Jasmolin I.Nomenclature
CAS number: 4466-14-2
(1R,3R)-2,2-Dimethyl-3-(2-methyl-1-propenyl)cyclopropanecarboxylic
acid (1S)-2-methyl-4-oxo-3-(2Z)-2-pentenyl-2-cyclopenten-1-yl
ester; 4′,5′-dihydropyrethrin I. C21H30O3; mol wt 330.46.
C 76.33%, H 9.15%, O 14.52%.
Properties
Liquid. uv max (Spectrosol hexane): 219 nm (ε 21500).Derivative
Jasmolin II.Nomenclature
CAS number: 1172-63-0
(1R,3R)-3-[(1E)-3-Methoxy-2-methyl-3-oxo-1-propenyl]-2,2-dimethylcyclopropanecarboxylic
acid (1S)-2-methyl-4-oxo-3-(2Z)-2-pentenyl-2-cyclopenten-1-yl
ester; 4′,5′-dihydropyrethrin II. C22H30O5; mol wt 374.47.
C 70.56%, H 8.07%, O 21.36%.