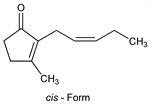
5259. Jasmone
Nomenclature
Description and references
Found in the volatile portion of oil from jasmine flowers. Natural jasmone is the cis-ketone. Isoln and structure: Ruzicka, Pfeiffer, Helv. Chim. Acta 16, 1208 (1933). Stereochemistry: Crombie, Harper, J. Chem. Soc. 1952, 869. Synthesis of cis-jasmone: H. Hunsdiecker, Ber. 75, 447 (1942); Stork, Borch, J. Am. Chem. Soc. 86, 936 (1964); Büchi, Wuest, J. Org. Chem. 31, 977 (1966); Sakan et al., Chem. Lett. 1973, 713; P. Bakuzis, M. L. F. Bakuzis, J. Org. Chem. 42, 2362 (1977). Synthesis of dihydrojasmone and cis-jasmone: C. S. Subramaniam et al., J. Chem. Soc. Perkin Trans. 1 1979, 2346; T. Kato et al., Chem. Pharm. Bull. 28, 349 (1980). Synthesis of trans-jasmone: Sisido et al., J. Org. Chem. 29, 2290 (1964). Comprehensive synthetic reviews: R. A. Ellison, Synthesis 1973, 397; T. L. Ho, Synth. Commun. 4, 265 (1974).