5267. Josamycin
Nomenclature
Description and references
Macrolide antibiotic produced by Streptomyces narbonensis var. josamyceticus nov. var. Isoln and characterization: T. Osono et al., J. Antibiot. 20A, 174 (1967). Manuf process: H. Umezawa,T. Osono, JP 66 21759 (1966 to Microbiochemical Res. Found.), C.A. 66, 54258w (1967). Prepn: Y. Oka et al., JP Kokai 76 41497 (1976 to Yamanouchi), C.A. 85, 121788b (1976). Identification with leucomycin A3: S. Omura et al., J. Antibiot. 23, 511 (1970). Structure: eidem, ibid. 27, 366 (1974). Absolute configuration: A. Ducruix et al., Chem. Commun. 1976, 947. Stereospecific total synthesis: K. Tatsuta et al., Tetrahedron Lett. 1980, 2837. Retrosynthetic studies: K. C. Nicolaou et al., J. Am. Chem. Soc. 103, 1222 (1981). A 17-membered aglycone was proposed at one time, see T. Osono, H. Umezawa in Drug Action and Drug Resistance in Bacteria 1, S. Mitsuhashi, Ed. (University Park Press, Baltimore, 1971) pp 41-120; T. Osono et al., J. Antibiot. 27, 366 (1974). Pharmacology: K. Kuriaki et al., Jpn. J. Antibiot. 22, 232 (1969). Review: T. Osono, H. Umezawa, loc. cit.
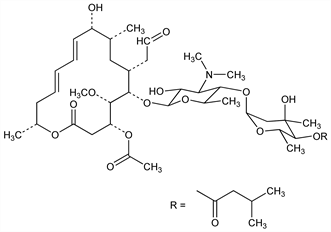
Properties
Colorless needles from benzene, mp 130-133° (after drying under reduced pressure at 100° for 5 hrs). [α]D25 70° (c = 1 in ethanol). uv max (0.001N HCl): 232 nm (E1%1cm 325). pKa 7.1 (40% aq methanol). Very sol in methanol, ethanol, acetone, chloroform, ethyl acetate, dioxane, acidic water. Sol in butanol, ether, CCl4, benzene, toluene. Practically insol in water, petr ether, ligroin, n-hexane.Derivative
Propionate.Nomenclature
Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); Macrolides