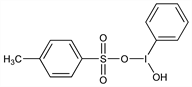
5321. Koser's Reagent
Nomenclature
CAS number: 27126-76-7
Hydroxy(4-methylbenzenesulfonato-κO)phenyliodine; [hydroxy(tosyloxy)iodo]benzene; HTIB; phenyliodoso hydroxide tosylate; phenylhydroxytosyloxyiodine. C13H13IO4S; mol wt 392.21.
C 39.81%, H 3.34%, I 32.36%, O 16.32%, S 8.18%.
Description and references
Unsymmetrical aryl λ3-iodane. Prepn: O. Y. Neiland, B. Y. Karele, J. Org. Chem. USSR 6, 889 (1970). Solid state structure determn by single-crystal x-ray analysis: G. F. Koser et al., J. Org. Chem. 41, 3609 (1976). Review of use in organic synthesis: R. M. Moriarty et al., Synlett 1990, 365-383; of chemistry: G. F. Koser, Aldrichim. Acta 34, 89-102 (2001).