5322. Kosins
Description and references
Phloroglucinols from flowers of Hagenia abyssinica J. J. Gmel. (Brayera anthelmintica Kunth.), Rosaceae. Isoln and early characterization: M. Leichsenring, Arch. Pharm. 232, 50 (1894); A. Lobeck, ibid. 239, 672 (1901); B. A. Hems, A. R. Todd, J. Chem. Soc. 1937, 562. Structure of α-kosin: W. Riedl, Ber. 89, 2600 (1956). Orginally identified as α- and β-kosins; ultimately shown to be a mixture of compds differing only in their acyl side chains. Structural elucidation of kosins and protokosin: M. Lounasmaa et al., Acta Chem. Scand. B28, 1200 (1974). Mass spectrometry: idem, P. Varenne, Planta Med. 34, 153 (1978). Identificaton and proportions in male and female flowers: T. Z. Woldemariam et al., Anal. Proc. 27, 178 (1990). Antitumor activity: idem, et al., J. Pharm. Biomed. Anal. 10, 555 (1992).
Derivative
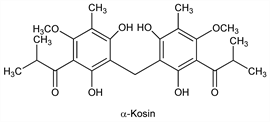
α-Kosin.Nomenclature
5,5′-Methylenebis[4,6-dihydroxy-2-methoxy-3-methylisobutyrophenone].Properties
Yellow needles from ethanol, mp 160-160.5°. uv max: 227, 290 nm (ε 30800, 24400). Soluble in alcohol, benzene, chloroform, ether, glacial acetic acid, alkalies.Derivative
Protokosin.Nomenclature
Description and references
Structurally related phloroglucinol. Exists in several tautomeric forms and is a mixt of isobutyryl, isovaleryl, and 2-methylbutyryl side chain homologues.
Properties
Colorless needles from acetone, mp 181-183°. [α]D25 +13.9° (c = 0.610 in chloroform). uv max (cyclohexane): 224, 285 nm (ε 28200, 36400). Practically insol in water. Slightly sol in alcohol, light petroleum; freely sol in ether, acetone, ethyl acetate, chloroform.Therapeutic Category
Has been used as anthelmintic (Cestodes).
Keywords
Anthelmintic (Cestodes)