5336. dl-Lactic Acid
Nomenclature
Description and references
Occurs in sour milk as a result of lactic acid bacteria; also found in molasses due to partial conversion of sugars, in apples and other fruits, tomato juice, beer, wines, opium, ergot, foxglove, and several higher plants, especially during germination. Lactic acid is prepd technically by “lactic acid fermentation” of carbohydrates such as glucose, sucrose, lactose with Bacillus acidi lacti or related organisms such as Lactobacillus delbrueckii, L. bulgaricus etc. The fermentation is carried out at relatively high temps. Produced commercially by fermentation of whey, cornstarch, potatoes, molasses. Review on the production of lactic acid by fermentation: S. C. Prescott, C. G. Dunn, Industrial Microbiology (McGraw-Hill, New York, 3rd ed., 1959) pp 304-331. Chem prepns from acetaldehyde and CO in dil H2SO4 at 130-200° and 900 atm: Loder, US 2265945 (1938 to du Pont); by hydrolysis of hexoses with NaOH: Lock, US 2382889 (1943). Prepn of crystalline lactic acid: Borsook et al., J. Biol. Chem. 102, 449 (1933). Toxicity data: Smyth et al., J. Ind. Hyg. Toxicol. 23, 259 (1941). Comprehensive description: F. J. Al-Shammary et al., Anal. Profiles Drug Subs. Excip. 22, 263-316 (1993).
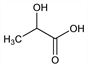
Properties
Crystals, mp 16.8°. bp14-15 122°; bp0.5-1 82-85°. pKa at 25° 3.86. nD20 1.4262. Heat of combustion at constant pressure 3615 cal/kg. Volatile with superheated steam. Sol in water, alc, furfurol; less sol in ether. Practically insol in chloroform, petr ether, carbon disulfide. Caustic in concd solns. LD50 orally in rats: 3.73 g/kg (Smyth).Derivative
Barium salt.Nomenclature
Properties
Powder. Poisonous. Sol in water, dil alcohol.Derivative
Copper salt dihydrate.Nomenclature
Properties
Green to blue crystals. Readily sol in water. Practically insol in alcohol.Use
In dyeing baths, as mordant in printing woolen goods, solvent for water-insoluble dyes (alcohol-soluble induline, nigrosine, spirit-blue). Reducing chromates in mordanting wool. Food preservative. Manuf cheese, confectionery. Component of babies' milk formulas; acidulant in beverages; for acidulating worts in brewing. In prepn of sodium lactate injections. Ingredient of cosmetics. Component of spermatocidal jellies. For removing Clostridium butyricum in manuf of yeast; dehairing, plumping, and decalcifying hides. Solvent for cellulose formate. Flux for soft solder. Manuf lactates which are used in food products, in medicine, and as solvents. Plasticizer, catalyst in the casting of phenolaldehyde resins.Therapeutic Category
Acidulant.