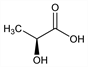
5337. l-Lactic Acid
Nomenclature
Description and references
Occurs in small quantities in the blood and muscle fluid of man and animals. The lactic acid concn increases in muscle and blood after vigorous activity. l(+)-Lactic acid is also present in liver, kidney, thymus gland, human amniotic fluid, and other organs and body fluids. Obtained by resolution of dl-lactic acid: Purdie, Walker, J. Chem. Soc. 61, 754 (1892); Borsook et al., J. Biol. Chem. 102, 449 (1933). Convenient laboratory prepn from glucose by fermentation by Lactobacillus delbrueckii: Brin, Biochem. Prep. 3, 61 (1953). Prepn from hexoses using B. dextrolacticus: Andersen, Greaves, Ind. Eng. Chem. 34, 1522 (1942). Monograph: M. Brin, R. H. Dunlop, “Chemistry and Metabolism of l- and d-Lactic Acids” in Ann. N.Y. Acad. Sci. 119, 851-1165 (1965).