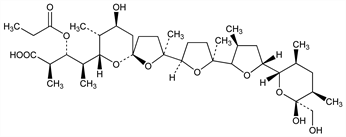
5348. Laidlomycin
Nomenclature
Description and references
Polyether ionophore antibiotic; structurally related to monensin, q.v. Isoln from Streptomyces spp. and antimicrobial activity: F. Kitame et al., J. Antibiot. 27, 884 (1974). Structure determn: F. Kitame, N. Ishida, ibid. 29, 759 (1976). Prepn of esters: A. F. Kluge, R. D. Clark, EP 24189 (1981 to Syntex). 13C-NMR study and enhancement of biological activities: R. D. Clark et al., J. Antibiot. 35, 1527 (1982). Mechanism of action study: U. Grfe et al., J. Basic Microbiol. 29, 391 (1989). HPLC determn in fermentation broth: M. Beran et al., Chromatographia 31, 603 (1991). Effect on cattle growth: M. T. Van Koevering et al., 1991 Animal Science Research Report (Agricultural Experiment Station, Oklahoma State University, Stillwater, OK, 1991) 241; M. L. Galyean et al., J. Anim. Sci. 70, 2950 (1992).