5383. Laureline
Nomenclature
CAS number: 81-38-9
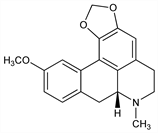
(7aR)-6,7,7a,8-Tetrahydro-11-methoxy-7-methyl-5H-benzo[g]-1,3-benzodioxolo[6,5,4-de]quinoline; 10-methoxy-1,2-(methylenedioxy)aporphine. C19H19NO3; mol wt 309.36.
C 73.77%, H 6.19%, N 4.53%, O 15.52%.
Description and references
From bark of Laurelia novae-zelandiae A. Cunn., Lauraceae: Aston, J. Chem. Soc. 97, 1381 (1910). Synthesis: Schlittler, Helv. Chim. Acta 15, 394 (1932); Faltis et al., Ber. 77B, 686 (1945); Gibson et al., J. Chem. Soc. C 1970, 2234; Govindachari et al., Indian J. Chem. 8, 475 (1970).
Properties
Cubes from petr ether. [α]D20 99.2° (c = 0.736 in 50% alc). Absorption spectrum: Girardet, J. Chem. Soc. 1931, 2636. Feebly basic. Readily oxidizes in air. Sol in alcohol, ether. Practically insol in water.Derivative
Hydrochloride.C19H19NO3.HCl; mol wt 345.82.
C 65.99%, H 5.83%, N 4.05%, O 13.88%, Cl 10.25%.
Properties
Crystals, mp 280°. [α]D20 57°. Sparingly sol in water.Derivative
(S)-Form.Nomenclature
CAS number: 65981-49-9
Properties
Cubes from petr ether, mp 114-115°. [α]D20 +97.9° (c = 1.12 in abs alc); [α]D20 +100° (c = 0.560 in CS2).Derivative
(±)-Form.Nomenclature
CAS number: 3749-97-1
Properties
Rough needles from petr ether, mp 115-116°.Derivative
Nitrate.C19H19NO3.HNO3; mol wt 372.37.
C 61.28%, H 5.41%, N 7.52%, O 25.78%.
Properties
Crystals, mp 238-240°. Sparingly sol in water.Derivative
Hydriodide.C19H19NO3.HI; mol wt 437.27.
C 52.19%, H 4.61%, N 3.20%, O 10.98%, I 29.02%.
Properties
Crystals. Practically insol in water.Derivative
Oxalate.(C19H19NO3)2.(COOH)2; mol wt 708.75.
C 67.79%, H 5.69%, N 3.95%, O 22.57%.