5459. Levamisole
Nomenclature
Description and references
Biological response modifier with anthelmintic activity. Prepn of racemate: A. H. M. Raeymaekers et al., US 3274209 (1966 to Janssen); eidem, J. Med. Chem. 9, 545 (1966). Pharmacology: D. Thienpoint et al., Nature 209, 1084 (1966). Prepn and abs config of isomers: A. H. M. Raeymaekers et al., Tetrahedron Lett. 1967, 1467. See also: M. W. Bullock et al., J. Med. Chem. 11, 169 (1968); US 3565907 (1971 to Am. Cyanamid); Dewar et al., US 3579530 (1971 to ICI). Stereospecific inhibition of succinic dehydrogenase: H. Van den Bossche, P. A. J. Janssen, Biochem. Pharmacol. 18, 35 (1969). Exptl effect on IL-1 production: E. S. Kimball, Ann. N.Y. Acad. Sci. 685, 259 (1993). Review of pharmacology: H. Schneiden, Int. J. Immunopharmacol. 3, 9 (1981); of clinical use in parasitic infections: M. J. Miller, Drugs 20, 122-130 (1980). Determn in animal tissues by ELISA: J. J. Silverlight, R. Jackman: Analyst 119, 2705 (1994); by LC-MS: A. Cannavan et al., ibid. 120, 331 (1995). Clinical trial with fluorouracil in colon cancer: C. G. Moertel et al., Ann. Intern. Med. 122, 321 (1995). Review of immunopharmacology and clinical trials in cancer treatment: H. C. Stevenson et al., J. Clin. Oncol. 9, 2052-2066 (1991); M. De Brabander et al., Anticancer Res. 12, 177-188 (1992); W. K. Amery, J. P. Bruynseels, Int. J. Immunopharmacol. 14, 481-486 (1992).
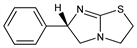
Properties
mp 60-61.5°. [α]D25 85.1° (c = 10 in chloroform).Derivative
Hydrochloride.Nomenclature
Properties
mp 227-229°. [α]D20 124 ± 2° (c = 0.9, water). Sol in water. Stable in aq acid media.Derivative
dl-Form.Nomenclature
Properties
Crystals, mp 87-89°.Derivative
dl-Form hydrochloride.Nomenclature
Properties
Crystals, mp 264-265°. Sol in water (21 g/100 ml at 20°), methanol, propylene glycol; sparingly sol in ethanol. Slightly sol in chloroform, hexane, acetone. LD50 in mice, rats (mg/kg): 22, 24 i.v.; 84, 130 s.c.; 210, 480 orally (Thienpoint).Derivative
d-(+)-Form.Nomenclature
Dexamisole.Properties
mp 60-61.5°. [α]D25 +85.1° (c = 10 in chloroform).Derivative
d-(+)-Form hydrochloride.Nomenclature
R-12563.Properties
mp 227-227.5°. [α]D20 +125° (c = 0.7 in water).Therapeutic Category
Anthelmintic (nematodes); immunomodulator.
Therapeutic Category (Veterinary)
Anthelmintic (Nematodes).Keywords
Anthelmintic (Nematodes); Immunomodulator