5460. Levcromakalim
Nomenclature
CAS number: 94535-50-9
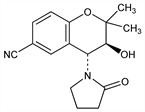
(3S-trans)-3,4-Dihydro-3-hydroxy-2,2-dimethyl-4-(2-oxo-1-pyrrolidinyl)-2H-1-benzopyran-6-carbonitrile; (3S,4R)-3-hydroxy-2,2-dimethyl-4-(2-oxo-1-pyrrolidinyl)-6-chromancarbonitrile; ()-6-cyano-3,4-dihydro-2,2-dimethyl-trans-4-(2-oxo-1-pyrrolidinyl)-2H-benzo[b]pyran-3-ol; lemakalim; ()-cromakalim; BRL-38227. C16H18N2O3; mol wt 286.33.
C 67.12%, H 6.34%, N 9.78%, O 16.76%.
Description and references
Potassium channel opener. Prepn of racemate: J. M. Evans et al., EP 76075; eidem, US 4446113 (1983, 1984 both to Beecham); of isomers: E. Faruk, EP 120428 (1984 to Beecham); V. A. Ashwood et al., J. Med. Chem. 29, 2194 (1986). Mechanism of action and hemodynamic effects: R. P. Hof et al., Circ. Res. 62, 679 (1988). Pharmacology: J. C. Clapham et al., Arzneim.-Forsch. 41, 385 (1991). Clinical evaluation in hypertension: S. Suzuki et al., ibid. 45, 859 (1995).
Properties
Crystals from ethyl acetate, mp 242-244°. [α]D26 52.2° (c = 1 in chloroform).Derivative
(+)-Form.Properties
Crystals from ethyl acetate, mp 243-245°. [α]D26 +53.5° (c = 1 in chloroform).Derivative
(±)-Form.Nomenclature
CAS number: 94470-67-4
Cromakalim; BRL-34915. Properties
Crystals from ethyl acetate, mp 230-231°.Therapeutic Category
Antihypertensive.
Keywords
Antihypertensive; Potassium Channel Activator/Opener