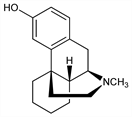
5470. Levorphanol
Nomenclature
Description and references
Orally active synthetic morphine analog. Prepn of racemate from 2-methyl-1-benzyl-1,2,3,4,5,6,7,8-octahydroisoquinoline: Grewe, Naturwissenschaften 33, 333 (1946); Angew. Chem. A59, 198 (1947); Grewe, Mondon, Ber. 81, 279 (1948); CH 280674 (1952 to Hoffmann-La Roche), C.A. 47, 7554 (1953). Prepn of isomers: Schnider, Grüssner, Helv. Chim. Acta 34, 2211 (1951); Vogler, US 2744112 (1956 to Hoffmann-La Roche). Absolute configuration: Corrodi et al., Helv. Chim. Acta 42, 212 (1959). Analgesic activity and toxicity data: L. O. Randall, G. Lehmann, J. Pharmacol. Exp. Ther. 99, 163 (1950). HPLC determn in plasma: R. Lucek, R. Dixon, J. Chromatogr. 341, 239 (1985). Clinical pharmacokinetics: R. Dixon et al., Res. Commun. Chem. Pathol. Pharmacol. 41, 3 (1983).
Properties
Crystals, mp 198-199°. [α]D20 56° (c = 3 in absolute alcohol).Derivative
Tartrate dihydrate.Nomenclature
Properties
Crystals, mp 113-115° (when anhydrous, mp 206-208°). [α]D20 14° (c = 3 in water). pH of a 0.2% aq soln 3.4 to 4.0. One gram dissolves in 45 ml water, in 110 g alcohol, in 50 g ether.Derivative
dl-Form.Nomenclature
Properties
Crystals from anisole and dil alcohol, mp 251-253°.Derivative
dl-Form hydrobromide.Nomenclature
Properties
Crystals, mp 193-195°. Sol in water; sparingly sol in alcohol. Practically insol in ether. LD50 i.v. in mice: 41 mg/kg (Randall, Lehmann).Derivative
d-Form.Nomenclature
Description and references
HPLC-MS/MS determn in urine: M. L. Constanzer et al., J. Chromatogr. B 816, 297 (2005).
Properties
Crystals, mp 198-199°. [α]D20 +56.3° (c = 3 in abs alcohol).Derivative
d-Form tartrate monohydrate.Properties
Crystals, mp 183-185°. [α]D20 +34.6° (c = 3 in water). Sol in water.Note
Levorphanol and racemorphan are controlled substances (opiates): 21 CFR, 1308.12.Therapeutic Category
Analgesic.
Keywords
Analgesic; Opioids; Morphinan Derivatives