5469. Levopropoxyphene
Nomenclature
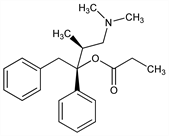
CAS number: 2338-37-6
(αR)-α-[(1S)-2-(Dimethylamino)-1-methylethyl]-α-phenylbenzeneethanol
propanoate (ester); α-l-4-dimethylamino-3-methyl-1,2-diphenyl-2-butanol
propionate; α-l-4-dimethylamino-1,2-diphenyl-3-methyl-2-butanol
propionate; l-propoxyphene. C22H29NO2; mol wt 339.47.
C 77.84%, H 8.61%, N 4.13%, O 9.43%.
Description and references
Prepn: Pohland, Sullivan, J. Am. Chem. Soc. 77, 3400 (1955). Stereoselective synthesis: Pohland et al., J. Org. Chem. 28, 2483 (1963). Toxicity data: E. I. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971). See also Propoxyphene.
Properties
Crystals from petr ether, mp 75-76°. [α]D25 68.2° (c = 0.6 in chloroform).Derivative
Hydrochloride.Nomenclature
CAS number: 1596-70-9
C22H29NO2.HCl; mol wt 375.93.
C 70.29%, H 8.04%, N 3.73%, O 8.51%, Cl 9.43%.
Properties
Crystals from methanol + ethyl acetate, mp 163-164°. [α]D25 60.1° (c = 0.7).Derivative
2-Naphthalenesulfonate.Nomenclature
CAS number: 5714-90-9
Levopropoxyphene napsylate; Novrad (Lilly); Letusin; Contratuss. C22H29NO2.C10H8SO3; mol wt 547.70.
C 70.17%, H 6.81%, N 2.56%, O 14.61%, S 5.85%.
Properties
LD50 orally in female rats: 1455 ±77 mg/kg (Goldenthal).Therapeutic Category
Antitussive.
Keywords
Antitussive