5497. Linamarin
Nomenclature
CAS number: 554-35-8
2-(β-d-Glucopyranosyloxy)-2-methylpropanenitrile; phaseolunatin. C10H17NO6; mol wt 247.25.
C 48.58%, H 6.93%, N 5.66%, O 38.83%.
Description and references
From the seed skins or embryos of flax: Jorissen, Hairs, Bull. Acad. Roy. Sci. Belg. [3] 21, 529 (1891); André et al., Compt. Rend. 231, 590 (1950); Lüdtke, Biochem. Z. 323, 428 (1953). Synthesis: Fischer, Anger, Ber. 52, 854 (1919). Biosynthesis in white clover: Butler, Butler, Nature 187, 780 (1960).
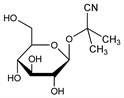
Properties
Bitter needles, mp 142-143°. [α]D18 29°. Freely sol in water, cold alcohol, hot acetone; slightly in hot ethyl acetate, ether, benzene, chloroform. Practically insol in petr ether. Evolves HCN with linseed meal but not with emulsin.Derivative
Tetraacetate.C18H25NO10; mol wt 415.39.
C 52.05%, H 6.07%, N 3.37%, O 38.52%.