5562. Lomefloxacin
Nomenclature
Description and references
Fluorinated quinolone antibacterial. DNA gyrase antagonist. Prepn: Y. Itoh et al., DE 3433924; eidem, US 4528287 (both 1985 to Hokuriku Pharm.). In vitro and in vivo activity: T. Hirose et al., Antimicrob. Agents Chemother. 31, 854 (1987). Comparative antibacterial spectrum in vitro: R. Wise et al., ibid. 32, 617 (1988). HPLC determn in urine and bile and preliminary pharmacokinetics in rats: A. Saito et al., ibid. 156. Photochemistry study: E. Fasani et al., Eur. J. Org. Chem.2004, 5075. Supplement on antibacterial spectrum, pharmacokinetics and clinical efficacy: Chemotherapy (Tokyo) 36, Suppl. 2, 1-1418 (1988). Comprehensive description: Y. D. Sanzgiri et al., Anal. Profiles Drug Subs. Excip. 23, 321-369 (1994).
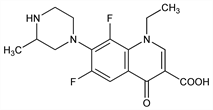
Properties
Colorless needles from ethanol, mp 239-240.5°. Soly at 25° (mg/ml): 1.03 at ionic strength 0.15 M NaCl. uv max (0.15 M acetate buffer, pH 5): 226, 288, 320 nm (aM × 103 15.5, 36.7, 13.5); uv max (0.05 M phosphate buffer, pH 7): 282, 326 nm (aM × 103 31.5, 13.3); uv max (0.15 M borate buffer, μ = 0.15 M with NaCl, pH 9): 282, 328 nm (aM × 103 29.3, 13.2). LD50 in mice (mg/kg): 245.6 i.v.; >4000 orally (Itoh).Derivative
Monohydrochloride.Nomenclature
Properties
Colorless needles from water, mp 290-300° (dec).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Synthetic); Quinolones and Analogs