5602. Lumisterol
Nomenclature
CAS number: 474-69-1
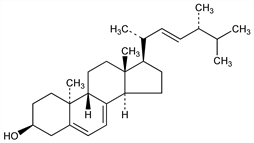
(3β,9β,10α,22E)-Ergosta-5,7,22-trien-3-ol. C28H44O; mol wt 396.65.
C 84.78%, H 11.18%, O 4.03%.
Description and references
Differs from ergosterol by spatial arrangement involving the methyl group at C-10. Prepd by ultraviolet irradiation of a benzene-alcohol soln of ergosterol: Askew et al., Proc. R. Soc. London B109, 488 (1932). Stereochemistry: Castells et al., J. Chem. Soc. 1959, 1159. Dehydrogenation with selenium gives Diel's hydrocarbon, 3′-methyl-1,2-cyclopentenophenanthrene.
Properties
Needles from acetone-methanol, mp 118°. [α]D19 +191.5°, [α]19546 +235.4° (c = 2 in acetone). uv max: 265, 280 nm: Windaus et al., Ann. 493, 259 (1932); Heilbron et al., J. Chem. Soc. 1937, 411. Practically insol in water. Sol in organic solvents. Forms a monomolecular compd with calciferol, mp 122°.Derivative
Acetate.C30H46O2; mol wt 438.69.
C 82.14%, H 10.57%, O 7.29%.