5603. Lunacridine
Nomenclature
CAS number: 83-58-9
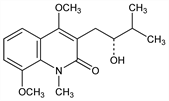
3-[(2R)-2-Hydroxy-3-methylbutyl]-4,8-dimethoxy-1-methyl-2(1H)-quinolinone. C17H23NO4; mol wt 305.37.
C 66.86%, H 7.59%, N 4.59%, O 20.96%.
Description and references
From bark of Lunasia costulata Miq., and L. quercifolia K. Schum. et Lauterb., Rutaceae. (+)-Form is naturally occurring. Isoln: Boorsma, Bull. Inst. Bot. Buitenzorg 21, 8 (1904). Structure: Goodwin, Horning, J. Am. Chem. Soc. 81, 1908 (1959). Synthesis: Clarke, Grundon, J. Chem. Soc. 1964, 438. Synthesis and stereochemistry: R. M. Bowman et al., J. Chem. Soc. Perkin Trans. 1 1973, 1051. Short synthesis of racemate: M. Ramesh, P. Shanmugam, Indian J. Chem. 24B, 767 (1985).
Properties
Crystals from methanol/water, mp 86-87°. [α]25589 +28.1°; [α]25436 +76.5° (c = 0.935 in ethanol). uv spectra: Goodwin, Horning, loc. cit. Very slightly sol in water; freely sol in alcohol, benzene, chloroform, ether, ethyl acetate, carbon disulfide, petr ether.Derivative
Hydroperchlorate.C17H24ClNO8; mol wt 405.83.
C 50.31%, H 5.96%, Cl 8.74%, N 3.45%, O 31.54%.
Properties
Shiny flakes from methanol/ether, mp 146-148°; remelts at 193-195°. [α]25589 +22.3°; [α]25436 +60.7° (c = 0.750 in ethanol).Derivative
(±)-Form.Nomenclature
CAS number: 52306-35-1