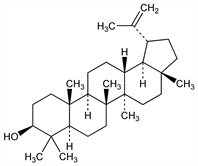
5608. Lupeol
Nomenclature
Description and references
Abundant plant triterpene. Occurs in the skin of lupin seeds, in chicle, in the latex of fig trees and of rubber plants. Was detected in cocoons of Bombyx mori. Isoln: Cohen, Rec. Trav. Chim. 28, 368 (1909); Ruzicka et al., Helv. Chim. Acta 20, 1567 (1937). Identity with β-viscol: Meyer, Jeger, ibid. 31, 1868 (1948). Identity with monogynol B: Chatterji et al., J. Sci. Ind. Res. 18B, 262 (1959). Structure: Ruzicka et al., Helv. Chim. Acta 28, 942 (1945); Ames et al., J. Chem. Soc. 1951, 450. Structure of hydrochloride: Halsall et al., ibid. 1952, 2862. Configuration: Barton, Holmes, ibid. 1952, 78; Djerassi et al., J. Am. Chem. Soc. 77, 5330 (1955). NMR studies: Buckley et al., Chem. Ind. (London) 1971, 298. Total synthesis: Stork et al., J. Am. Chem. Soc. 93, 4945 (1971).