5607. Lupanine
Nomenclature
(7α,7aα,14α,14aβ)-Dodecahydro-7,14-methano-2H,11H-dipyrido[1,2-a:1′,2′-e][1,5]diazocin-11-one; dodecahydro-7,14-methano-4H,6H-dipyrido[1,2-a:1′,2′-e][1,5]diazocin-4-one.Description and references
Racemic lupanine is found in white lupins, d-lupanine is found in blue lupins, l-lupanine has been prepd from the natural racemic form. Structure: Davis, Arch. Pharm. 235, 199, 218, 229 (1897); Clemo, Leitch, J. Chem. Soc. 1928, 1811; Clemo et al., ibid. 1931, 429; 1933, 644; Ing, ibid. 1933, 504; Karrer et al., Helv. Chim. Acta 11, 1062 (1928), 13, 1292 (1930); Winterfeld, Holschneider, Ber. 64, 137, 2415 (1931), 66, 1751 (1933); Ann. 499, 109 (1932); Winterfeld, Kneuer, Ber. 64, 150 (1931); Winterfeld, Hoffman, Arch. Pharm. 275, 5, 65 (1937). Abs config of l-form: Okuda et al., Chem. Ind. (London) 1961, 1116. Crystal structure of dl-form: H. Doucerain et al., Acta Crystallogr. B32, 3213 (1976). Biosynthetic study of d-form: W. M. Golebewski, I. D. Spenser, J. Am. Chem. Soc. 106, 7925 (1984). See also sparteine, q.v.
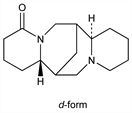
Derivative
dl-Form.Nomenclature
Properties
Orthorhombic prisms from acetone, mp 98-99°. bp1.0 185-195°. Sol in water, alcohol, ether, chloroform. Insol in petr ether. See: Soldaini, Arch. Pharm. 231, 321 (1893); Couch, J. Am. Chem. Soc. 56, 1423 (1934).Derivative
Dihydrochloride.Properties
Deliquescent prisms, mp 185° (dec).Derivative
Hydrochloride dihydrate.Properties
Crystals, mp 177-178°. mp 250-252° (dry).Derivative
d-Form.Nomenclature
Properties
Syrup crystallizing difficultly in hygroscopic needles, mp 40-44°. bp3 190-193°. nD24 1.5444. [α]D25 +84° (c = 4.8 in alc). Freely sol in water, alcohol, chloroform, ether. Sol in petr ether.Derivative
Hydrochloride dihydrate.Properties
Rhombic crystals from water, mp 127° (dry).Derivative
l-Form.Nomenclature
Properties
Viscous oil, bp1.0 186-188°. [α]D about 61° in acetone.Derivative
α-Isolupanine.Nomenclature
Description and references
cis-cis-Isomer of lupanine: Marion, Leonard, Can. J. Chem. 29, 355 (1951). Isoln of d-form from Lupinus sericeus Pursh., L. perennis L. and L. augustifolius, Leguminosae: Marion et al., ibid. 31, 181 (1953); Rink, Schfer, Arch. Pharm. 287, 290 (1954); Winterfeld, Pies, ibid. 290, 537 (1957).